Part 5 - Fear and Redemption
How much clarity can a billion dollars buy?
Estrogen Is All You Need
Every year, a million American women, most in their 40s and 50s, enter perimenopause. Their sleep degrades. Their energy declines. They are overcome by waves of heat. But, what if I told you this condition was curable?
“Menopause is curable. Under proper treatment, nearly all symptoms cease in the vast majority of cases. The bodily changes typical of middle age can be reversed, and sexual functions can be restored, along with a fully feminine appearance.”1
In fact, it gets better! Menopause is not just curable, but preventable entirely!
“Menopause is completely preventable. No woman need suffer menopause or any of its symptoms if she receives preventive treatment before the onset of menopause.”2
Surely, there must be some trade-off here? No woman has made it into her 70s without going through menopause. Of course we now know that loss of ovarian function is what drives menopause. But, what’s the catch?
“There is increasing evidence that estrogen has a preventive effect on breast and genital cancers. In 1962, the Journal of the American Medical Association reported on one of my studies, in which a group of 304 women, aged forty to seventy, had been treated with estrogen for periods up to twenty-seven years. Given normal odds, as established by medical statistics, eighteen cases of cancer—either of the breast or the uterus—would normally be expected in this group. Instead, not a single case of cancer occurred!”3
What a miracle molecule! Estrogen will keep you young! And healthy! Perhaps estrogen is all you need? This incredible news came not from the unsavory pharmaceutical industry. Rather, this was the gospel of Robert Wilson. A community gynecologist, Wilson preached the parables of all-natural (equine) estrogen. His bible, Feminine Forever, was a New York Times best-seller. He was printed in Vogue, Newsweek, and Science Digest. He was eminently qualified—his bestseller dedicates two pages to his degrees, membership, and awards.4 It had to be true… Right?
And yet, it wasn’t. The gospel was largely baseless. In fact, self-reported clinical results compose the near-totality of Wilson’s published work. A collection of case reports and anecdotes, or “anecdotal data” if you prefer. It was pretty straight forward to observe the symptomatic benefits from adoption and withdrawal of estrogen. These effects are large, consistent, and reproducible. However, evidence of longer term efficacy and safety from randomized clinical studies was absent.
But, Wilson was selling a solution to a real problem. In the mid 1960s, much like today, most women go through menopause without any treatment. But, Feminine Forever, with some undisclosed financial support from estrogen manufacturer Ayerst, changed that. By 1975, estrogen prescriptions had doubled. The market for menopause therapy quadroupled.5 Patients wanted to believe the promise. Some estimates put the prevalence of estrogen adoption north of 50% in cities by 1975.6 What followed was a half-century reckoning: with cancer, with statistical rigor, and with the question of what happens when medicine promises more than it knows.
Part 4 of this series traced how estrogen rose from a laboratory curiosity to a cultural phenomenon: Premarin to Feminine Forever. Part 5 asks what happened when that phenomenon collided with evidence.
A Series of Safety Scares
One key selling point was safety. Not only was Ayerst’s Premarin estrogen effective, it was natural! Wilson was simply replacing a missing molecule, albeit with its equine metabolites. Wilson's vision had a philosophical elegance. “Estrogen therapy doesn't change a woman," he wrote. "On the contrary: it keeps her from changing.”7 The medication restored the natural state. Menopause was pathology. So, you’re safer with it than without it, right?
Patients were rightfully concerned. Not long before, a prior popular estrogen mimic, Diethylstilbestrol (DES) had led to cancers. First synthesized in 1938 by Edward Charles Dodds, DES was a structurally simple non-steroid molecule. That made it a cheap, potent, and orally active alternative to human-derived estrogen preparations. Dodds chose not to patent DES, so scores of companies manufactured it freely. The FDA approved DES in 1941, and by the mid-1940s physicians were prescribing it to pregnant women to prevent miscarriage. No clinical trials ever legitimated the practice. Yet, up to two million American women took DES during pregnancy.8
In 1971, Arthur Herbst and colleagues at Massachusetts General Hospital published a devastating NEJM report: a cluster of young women with clear-cell vaginal adenocarcinoma, an extraordinarily rare cancer, were all daughters of women who had taken DES during pregnancy.9 The drug was pulled from obstetric use. Subsequent research found additional adverse effects in exposed sons and daughters, plus increased breast cancer risk in the mothers themselves.10
Intriguingly, the most concerning side-effect of DES didn’t emerge for decades, and in the next generation no less. But, from origin as a structurally distinct synthetic estrogen mimetic, rumors of DES-lined side-effects swirled for decades. The prevailing bias favored natural estrogens, like Premarin. The natural-versus-synthetic distinction became a commercial asset. DES was artificial and dangerous; Premarin was natural and, therefore, presumably safe. The assumption persisted largely unexamined until the mid-1970s.
Endometrial Cancer Emerges
On December 4, 1975, the New England Journal of Medicine published two studies that shook the estrogen consensus. Two independent teams had arrived, separately, at the same conclusion: unopposed estrogen dramatically increased the risk of endometrial cancer.
Smith et al. compared 317 women with endometrial cancer to 317 controls and found that estrogen users faced a 4.5-fold greater risk. Women without typical predisposing factors who took estrogen had the highest likelihood of developing the disease.11 Ziel and Finkle using Kaiser Permanente records found a duration-dependent relationship: risk was up to fourteen times higher for women who took conjugated estrogens for more than seven years.12 Both studies were published alongside two commissioned editorials — an extraordinary editorial decision underscoring the gravity the journal’s editors attached to the findings.23
Corroboration came rapidly. By 1979, at least six additional studies confirmed the link, with relative risks ranging from 3.1 to 15, all showing dose- and duration-dependence. A natural experiment in Seattle provided the most elegant evidence: as estrogen prescriptions declined in the late 1970s, endometrial cancer incidence declined in parallel.13
According to historian Elizabeth Watkins, the medical profession did not immediately capitulate. For example, Robert Kistner of Harvard Medical School dismissed the epidemiological evidence, attributing elevated cancer rates to misdiagnosis and concluding, memorably, that
‘‘From studying the effects of estrogen, progesterone, and other progestins on the endometrium in animals and women for 20 years, I am convinced that estrogens per se are not a cause of endometrial cancer in the human female… endometrial cancer is a disease of those women who have easy access to physicians.” (Watkins 2007 p. 99)
Two Yale biostatisticians, funded in part by Ayerst Laboratories, published a rebuttal claiming the risk was only 1.7-fold, but the NEJM editors published an editorial dismantling their methodology in the same issue. By 1979, however, “the pendulum had swung decisively in favor of a causal relationship between prescription estrogen and endometrial cancer.”14
The market responded immediately. According to Watkins, “In 1976 alone, dollar sales for menopausal estrogens decreased 11 percent and the number of prescriptions dropped 19 percent.”15 Feminists of the era, some skeptical of hormone therapy for a plethora of reasons, argued
“Estrogen replacement therapy is dangerous. It will raise your cancer risk. It may lead to vascular disease. It may even kill you.” (Rosetta Reitz, Menopause: A Positive Approach. 1977 from Houck 2003 p. 119)16
Adding Progestin
The endometrial cancer crisis was resolved by adding synthetic progesterone to the regimen. Robert Greenblatt proposed combining estrogen with progestin for five to seven days per cycle “to prevent endometrial carcinoma by causing the uterus to shed its lining.”17 The fix worked remarkably well. By the early 1980s, it became standard protocol, and multiple studies showed that “the addition of progesterone... not only negated the increased risk of uterine cancer associated with estrogen alone but actually protected against endometrial cancer; women receiving progesterone with estrogen had a lower incidence of endometrial cancer than women who received no hormones.”18
It is worth pausing on what medicine chose not to do. Some physicians proposed an alternative solution: prophylactic hysterectomy. Remove the uterus and you remove the cancer risk, freeing the patient for continued estrogen therapy. Hysterectomy was incredibly popular at the time. By the mid-1970s, nearly half of post-menopausal women underwent a hysterectomy. Watkins observes, with understated horror, that this:
“reveals a curious disregard for the inherent value of a postmenopausal woman’s uterus… indicates the extent to which menopause had become medicalized: these doctors did not question the widespread practice of major abdominal surgery among older women, but instead condoned the procedure as part of the risk-benefit calculus for estrogen replacement.”19
The progestin compromise resolved the endometrial cancer problem but introduced a new variable whose own risks, particularly regarding breast cancer and cardiovascular events, would not be understood for decades. Combined HRT (estrogen plus progestin) became standard for women. The combination of conjugated equine estrogens (CEE) plus medroxymedroxyprogesterone acetate (MPA), specifically Prempro, that would later be tested in the Women’s Health Initiative.
Meanwhile, the breast cancer question was accumulating; slowly, ambiguously, and without the decisive clarity of the endometrial cancer data. Pre-1985 studies produced inconsistent results; post-1985 studies largely agreed that long-term menopause hormone therapy increased breast cancer risk. Two dueling meta-analyses in 1991 reached opposite conclusions: a Vanderbilt analysis found negligible risk at standard doses (RR 1.08); a CDC/Emory analysis estimated that long-term estrogen use “could be responsible for at least 4,708 new cases and 1,468 deaths from breast cancer each year.”20 By 1992, the situation was untenable.
One thing, however, was becoming clear: observational studies could not resolve the question. The “healthy user” bias contaminated every non-randomized comparison. The call for a large randomized controlled trial became a chorus. That call would culminate in the Women’s Health Initiative.
Realizing Rigor
The Women’s Health Initiative was born from a specific political and scientific moment: the arrival of women in positions of institutional power, and recognition that decades of prescribing rested on an evidence base that could not answer the key questions.
Bernadine Healy, confirmed in 1991 as the first female director of the National Institutes of Health, used her position as a bully pulpit. At Senate hearings on women’s health, she announced the launch of what she called “the largest community-based clinical, prevention and intervention trial ever conducted... to examine the major causes of morbidity and mortality in women of all races and all socioeconomic strata.”21 In a companion NEJM editorial titled “The Yentl Syndrome,” she documented sex bias in cardiac care: women received less aggressive treatment than men unless they presented with symptoms matching the male profile of heart disease.22 Medicine, she argued, could no longer treat women as a smaller variant of men.
The WHI enrolled 161,809 postmenopausal women (aged 50–79) across the country. The estrogen-progestin arm randomized 16,608 women to either Prempro or placebo. The study cost nearly one billion dollars in 1992. It was “big science” arriving in women’s health: a Manhattan Project for menopause.
But even before the WHI reported, the HERS trial (Heart and Estrogen/Progestin Replacement Study) delivered a warning shot. Funded by Wyeth-Ayerst and completed in 1998, HERS found that combined HRT did not reduce heart attacks in women with existing heart disease and nearly tripled the risk of blood clots.23 The investigators were so shocked that they asked Wyeth to verify whether the hormone and placebo pills had been accidentally switched. Yet, HERS barely dented the estrogen consensus. Prescriptions continued to rise. Even Bernadine Healy, after HERS, continued to recommend estrogen for cardiac prevention on CBS Evening News.24 It would take the WHI’s sheer scale and dramatic termination to break through.
July 2002: the end of MHT?
In May 2002, the WHI’s Data and Safety Monitoring Board, reviewing the data for the tenth time, found that breast cancer diagnoses among participants had exceeded the predetermined safety threshold. The estrogen-progestin arm was stopped three years early.
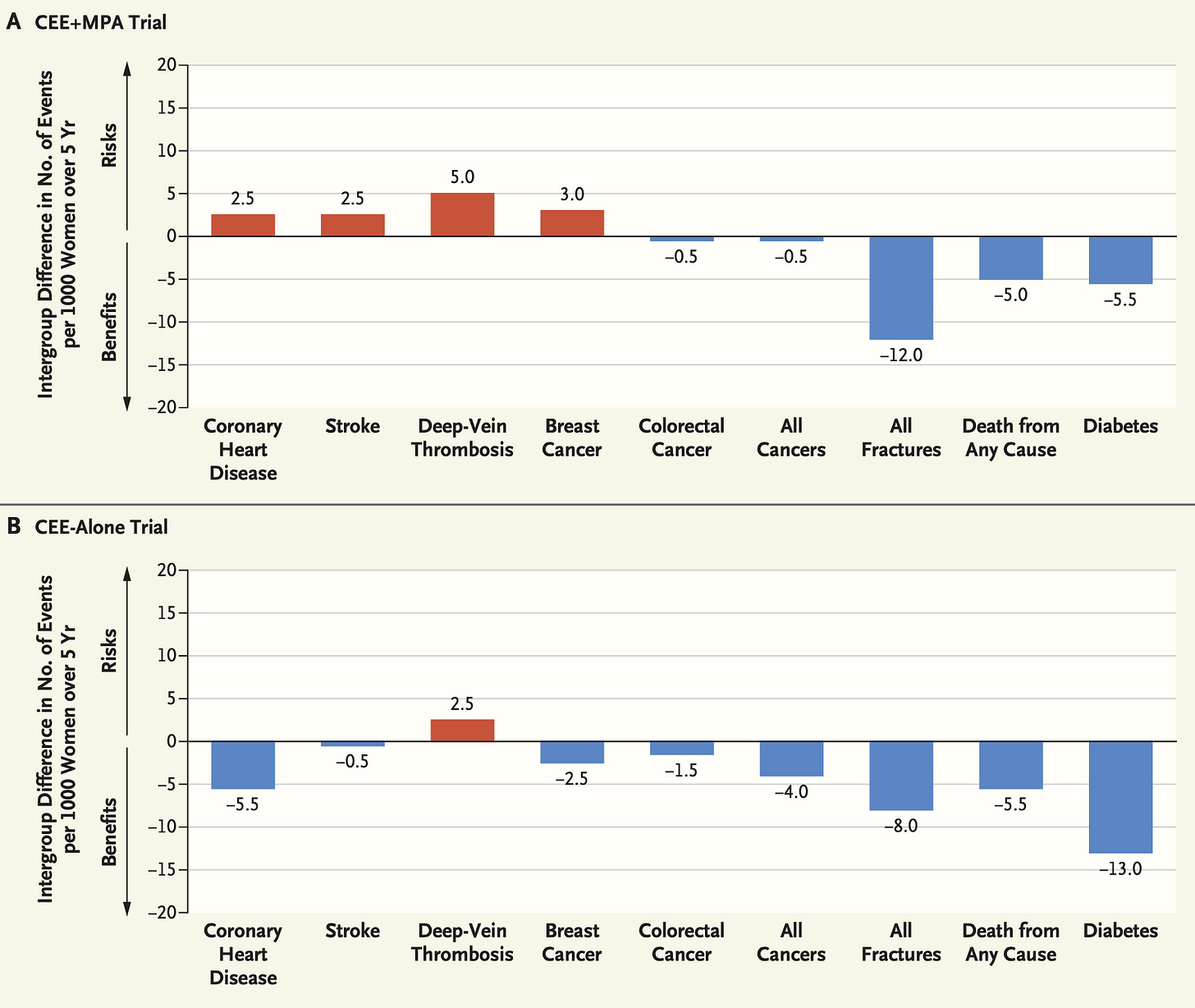
The results, expressed in relative terms: women on Prempro had a 26 percent increase in breast cancer (HR 1.26), a 29 percent increase in coronary heart disease (HR 1.29), a 41 percent increase in stroke (HR 1.41), and a 113 percent increase in pulmonary embolism (HR 2.13). There were benefits too: a 37 percent reduction in colorectal cancer and a 34 percent reduction in hip fracture. In absolute terms, these numbers translated to 8 more breast cancers, 7 more coronary events, 8 more strokes, and 8 more pulmonary embolisms per 10,000 women per year alongside 6 fewer colorectal cancers and 5 fewer hip fractures.25
JAMA fast-tracked the article. NIH issued a press release. Letters went to 16,000 participants instructing them to stop their pills. Headlines: “Study Says Halt Hormone Therapy.” “Health Risk to Women Halts Hormone Study.” Within a year, Prempro prescriptions dropped 33 percent, Premarin’s by 66 percent, and total HRT prescriptions by more than a third.26
There was, however, a second arm of the study, the estrogen-only arm, for women who had previously undergone hysterectomy. When its results arrived in 2004, they told a significantly different story. No increased breast cancer risk. In fact, a 23 percent reduction in breast cancer (HR 0.77). Reduced hip fractures. The increased stroke risk remained, but the overall picture was far more favorable than the combination arm.27 This discrepancy suggested that the progestin, a synthetic progesterone derivative, medroxyprogesterone acetate (MPA), rather than the estrogen, might have been the primary culprit in the combination arm’s adverse findings. The distinction that would become central to the arguments that followed.
The WHI was a landmark study. It answered the question of whether combined estrogen-progestin therapy prevents heart disease with a definitive no. But its design, its communication, and its over-generalization introduced distortions that took more than a decade to correct.
The problems were not trivial. The median age of WHI participants was 63, more than a decade past the typical onset of menopause. The study did not test the population most likely to use HRT: women in their late 40s and 50s experiencing menopausal symptoms.
The WHI tested a single regimen oral CEE+MPA. The results were generalized to all women, all ages, all MHT formulations, and all routes of administration. The 2026 Makary JAMA Viewpoint calls this “a sweeping overreach that... was never warranted by the data.”28
The 26 percent increase in breast cancer the number that drove the headlines, drove the panic, and drove millions of women off their medications “almost reached nominal statistical significance.”29 Almost means it did not. Bluming and Tavris note the exchange at a 2002 continuing medical education conference, when a physician questioned the WHI investigator about the non-significant confidence interval. The investigator’s reply:
“What happens is, if it’s an important question and if it’s a big study... and you can’t do it again because it costs too much money, then they’ll say that’s the best data there is and then the statistical police have to leave the room.”30
Reanalysis: the return of MHT?
The reassessment, when it came, centered on a question the WHI had not been designed to answer: does the timing of hormone initiation matter?
In 2013, Manson et al. published a comprehensive age-stratified reanalysis of WHI data and the results were striking.31 For women aged 50–59 — the clinically relevant population — the risk-benefit balance was fundamentally different from the overall WHI findings. Fewer coronary events. Fewer cancers. Fewer deaths. Fewer fractures. Only modest increases in thrombosis and stroke. In 2016, Manson and Kaunitz published an NEJM perspective arguing that “our understanding of [menopause hormone therapy’s] benefits and risks has never been clearer” and that the absolute risk of adverse outcomes was “much lower in younger women than in older women.”32
The “timing hypothesis” that estrogen may be broadly protective when initiated near menopause but harmful when initiated decades later gained substantial support. It took until February 2026 for these findings to reach the drug label. FDA Commissioner Marty Makary, in a JAMA Viewpoint, argued that the original boxed warnings were “based on a misapplication of WHI results” and that the WHI’s specific findings about one regimen in one age group had been wrongly generalized to all women, all ages, and all formulations.33 The FDA removed cardiovascular disease, breast cancer, and probable dementia risk statements from the boxed warning retaining only the endometrial cancer warning for estrogen-alone products in women with intact uteruses.34
The chilling effect of the original warnings had been severe. In 2020, less than 5% of the women aged 46–65 received an HRT prescription. Modern formulations transdermal estradiol, which bypasses hepatic first-pass metabolism and is thought to reduce clotting risk, and oral micronized progesterone, which appears to have neutral effects on breast tissue, bear little resemblance to the oral CEE plus MPA regimen the WHI tested. The labeled recommendation now advises starting HRT within 10 years of menopause onset or before age 60.
Some WHI investigators have pushed back. Jaques Rossouw, a WHI lead investigator, argued that the FDA’s relabeling is a step backwards into the pre-WHI era and that no new large RCTs had been conducted to change the balance of known risks.
Did we learn anything?
First, acting without evidence is costly in both directions. The Wilson era demonstrated the danger of widespread prescribing based on physiological reasoning and anecdote. The post-WHI era demonstrated the equal and opposite danger: an overcorrection so severe that it denied therapy to the very women most likely to benefit. Both errors harmed women. The former by exposing them to unquantified risks. The latter by subjecting them to years of treatable suffering. Striking a balance here is hard. Only high quality clinical evidence can clarify. We will have to deal with the consequences of largely denying a generation of women access to low-risk menopause hormone therapy for the decades to come. We don’t understand the impacts yet, but I suspect we’ll see more diabetes and broken hips than we did in prior generations.
Second, statistical literacy. The WHI’s 26 percent increase in breast cancer, reported in relative terms, generated international panic. In absolute terms, it meant eight additional cases per 10,000 women per year. It’s a meaningful difference, especially over a lifetime, but the risk might be worth it during the perimenopausal years.
Third, the distance between a molecule and a medicine. Almost every medicine is a molecule, but few molecules prove to be medicines. Part 4 of this series ended with the observation that the distance between a molecule that works and a molecule that is safe is often measured in decades and tragedies. The pattern recurs: a plausible mechanism, observational enthusiasm, commercial amplification, belated RCT testing, overcorrection. It is not unique to estrogen.
Fourth, marketing blocks the generation of the evidence patients need. Once molecules are adopted en masse, resolving causality in the absence of massive increases in risk is difficult. Manufacturers have no incentive to risk the sales of their established products. For estrogen, it took favorable political winds, a billion dollars, and the largest clinical research endeavor in NIH history to get a cursory answer to the safety and efficacy questions. We can argue over the formulations, but CEE/CEE+MPA were the popular forms of the era. These studies should have been run pre-approval. The story of estrogen in Menopause Hormone Therapy is, if anything, an argument for a stronger FDA with more stringent pre-approval evaluation.
I can’t help but think of the enthusiasm for “natural” “peptides” like BPC-157, a 12-amino acid which matches no known human protein yet is marketed breathlessly as a cure for musculoskeletal aches and pains. Salesmen, often less qualified than Wilson but similarly untethered from reality, urge patients—often struggling with major unmet medical needs—to try injecting some of the latest and greatest white powders from China.35 Others sell a “peptides” as an intervention for health optimization: getting older? Take some TB-500 and you’ll feel like yourself again. The evidence in favor almost always boils down to a mixture of anecdotes and “trust me, bro.”
Do these “peptides” work? Are they safe? Who knows! Some of these “peptides” might be medicines in the future. One popular peptide, CJC-1295, a Growth Hormone Releasing Hormone fragment, is a synthetic derivative of an endogenous protein that was being developed by ConjuChem Biotechnologies. In fact, some popular peptides like “Reta” (retatrutide) are currently being developed by pharmaceutical companies and will soon be FDA approved. This is the best case scenario.
But, history tends to rhyme. The FDA is re-scheduling certain “peptides” for compounding. Soon, compounding pharmacies will be selling uninspected, (hopefully) white powders from China for mass consumption. Apparently “compounders” and online not-a-pharmacy sites can market unapproved products without any evidence. I’m not sure that’s a good idea for any involved. But, like with Ayerst and Wilson, I’m sure we’ll see financial relationships emerge between the makers and the boosters. Is that good for patients?
And, for the “peptide” boosters reading this, unless you want your favorite peptide to end up like estrogen—with perpetually vexed safety and efficacy—I recommend you find a way to pony up the cash and run the RCTs patients deserve. The cost of moving fast on weak evidence here is the long-term health and safety of patients. The risks of estrogen, CEE+MPA, and DES didn’t emerge until after years, sometimes decades, of exposure. Let’s not repeat the mistakes made with estrogen. The joy of being human is learning from the experience of others. Let’s make our mothers proud.
Last, separate evaluation from origination. Perhaps the most discomfiting lesson is the one embedded in the ethical debates of the 1930s, traced in Part 4. The conservative physicians who urged restraint—low dose, short duration, symptoms only—turned out to be roughly correct. But they were correct for the wrong reasons: not because they had evidence of harm, but because they believed menopause was a natural verdict that women should accept. The physicians who later championed lifelong estrogen, Masters, Shelton, and Wilson, were more sympathetic to women’s suffering and more willing to take their preferences seriously. They were also completely untethered to the evidence for its safety and efficacy. Good intentions, poor evidence, reputational esteem, and aggressive promotion made a more harmful combination than paternalistic caution. The best way to separate the ideas from the baggage: randomized, double-blind placebo controlled clinical trials.
Coda
If the FDA’s 2026 relabeling represents a genuine correction, an evidence-based acknowledgment that the post-WHI overcorrection went too far, then medicine has, in some sense, learned from this story. The new labels are conditional. They specify age ranges. They distinguish formulations. They trade the blunt instrument of a boxed warning for the finer grain of clinical guidance. That looks like progress.
But if “remove the black box” becomes the next Feminine Forever — if the pendulum swings past the evidence again, propelled by commercial enthusiasm and a cultural appetite for pharmaceutical solutions for aging — then the lesson has been lost
The difference between the two will be measured not in labels, but in evidence. And the evidence, as Bernadine Healy understood when she launched the WHI, takes time and money. I don’t think the estrogen story is over. If anything, it is, for the moment, better informed.
Wilson, R.A. (1966). Feminine Forever. New York: M. Evans, p. 18 (Introduction).
Wilson, Feminine Forever, p. 19
Wilson, Feminine Forever, p. 67 (Chapter 3: "A Woman's Chemistry")
Wilson, Feminine Forever, p. 211-212
Watkins, E.S. (2007). The Estrogen Elixir: A History of Hormone Replacement Therapy in America. Johns Hopkins UP, p. 49
Watkins, The Estrogen Elixir, p. 49
Wilson, Feminine Forever, p. 53
Watkins, Estrogen Elixir, pp. 24–26
Herbst, A.L., Ulfelder, H., and Poskanzer, D.C. (1971). "Adenocarcinoma of the Vagina — Association of Maternal Stilbestrol Therapy with Tumor Appearance in Young Women." NEJM 284: 878–81. DOI: 10.1056/NEJM197104222841604
Watkins, Estrogen Elixir, p. 26
Smith, D.C. et al. (1975). “Association of exogenous estrogen and endometrial carcinoma.” NEJM 293: 1164–67; Watkins, Estrogen Elixir, pp. 93–94
Ziel, H.K. and Finkle, W.D. (1975). “Increased risk of endometrial carcinoma among users of conjugated estrogens.” NEJM 293: 1167–70; Watkins, Estrogen Elixir, pp. 93–95
Watkins, Estrogen Elixir, pp. 96–107
Watkins, Estrogen Elixir, pp. 101–107
Watkins, Estrogen Elixir, p. 104
Houck, J.A. (2003). "What Do These Women Want?" Bull. Hist. Med. 77(1): 117
Watkins, Estrogen Elixir, p. 105
Bluming, A. and Tavris, C. (2018). Estrogen Matters. Little, Brown, p. 18
Watkins, Estrogen Elixir, p. 101
Watkins, Estrogen Elixir, pp. 215–216
Watkins, Estrogen Elixir, p. 227
Healy, B. (1991). “The Yentl Syndrome.” NEJM 325: 274–76
Hulley, S. et al. (1998). "Randomized trial of estrogen plus progestin for secondary prevention of coronary heart disease in postmenopausal women." JAMA 280: 605–13; Watkins, Estrogen Elixir, pp. 265–267
Watkins, Estrogen Elixir, p. 268
Writing Group for the WHI Investigators (2002). “Risks and benefits of estrogen plus progestin in healthy postmenopausal women.” JAMA 288: 321–33; Watkins, Estrogen Elixir, p. 271.
Watkins, Estrogen Elixir, p. 264.
Women’s Health Initiative Steering Committee (2004). “Effects of conjugated equine estrogens in postmenopausal women with hysterectomy.” JAMA 291: 1701–12
Makary, M.A. et al. (2026). “Updated Labeling for Menopausal Hormone Therapy.” JAMA 335(2): 117–18
Bluming and Tavris, Estrogen Matters, pp. 33–34
Bluming and Tavris, Estrogen Matters, pp. 33–34
Manson, J.E. et al. (2013). “Menopausal hormone therapy and health outcomes during the intervention and extended poststopping phases of the WHI randomized trials.” JAMA 310: 1353–68
Manson, J.E. and Kaunitz, A.M. (2016). "Menopause Management — Getting Clinical Care Back on Track." NEJM 374(9): 803–6
Makary et al. (2026), JAMA 335(2): 117–18
Makary et al. (2026); FDA Press Release, February 12, 2026
One obvious objection: why is eating concentrated horse urine extract okay, but injecting white powders from a peptide printer in China is not? Unsatisfactory answer: eating is safer than injecting. And, a lot of women ate a lot of horse urine extract for a long time. It could have been dangerous - and some would argue was - but, it’s hard to ignore millions of people for decades. That said, I encourage the men thinking about injecting white powders to ask themselves how they’d feel on Premarin and if that feeling is different from their mothers.