Part 1: Reviving Estrogen Replacement Therapy
Recently, the FDA updated labeling of estrogen products for menopause. Why?
On February 12th, the US FDA made good on its promise to revise the labeling of Menopausal Hormone Therapy (MHT). These changes to focus on clarifying the risks from estrogen-based MHT that millions of peri- and post-menopausal women in the US use for symptoms associated with menopause. From the 1960s through the 1990s, MHT was a popular regiment to treat menopause-associated symptoms. However, after a series of large randomized controlled trials identified risk signals in the mid-2000s, the prevalence of MHT use among peri- and post-menopausal women in the US declined ≥80%, from 26.9% in 1999 to 4.7% in 2020.
In essence, a generation of women went through menopause without MHT support. And, a generation of clinicians trained in a practice that eschewed MHT for menopause. Can a change in labeling expand access and improve quality of life for patients?
A Transition
Menopause is the permanent cessation of menstruation resulting from the loss of ovarian follicular activity. It is diagnosed retrospectively, after 12 consecutive months without a menstrual period. The average age of onset in the United States is 51.1 But menopause is not an event; it is a transition. Functionally, menopause symptoms emerge as a consequence of ovarian ‘failure.’ The perimenopausal years preceding it are marked by rapid hormonal fluctuations as the ovary’s pool of follicles dwindles. The decline in estradiol, the principal estrogen produced by the premenopausal ovary, proceeds unevenly. The hypothalamic-pituitary axis, sensing the shortfall, ramps up gonadotropin secretion in an increasingly futile attempt to stimulate follicles that are no longer there. The result is a rapidly fluctuating hormone profile resulting from diminished feedback from the follicle. While humans are not uniquely capable of experiencing menopause, it does appear that a extended post-reproductive life period is relatively rare across the animal kingdom with toothed whales and chimpanzees as the best documented examples.2 So, how does this transition manifest for patients?
The clinical consequences of menopause are familiar to nearly every woman who reaches middle age. Nearly 75% of menopausal women experience vasomotor symptoms: hot flashes and night sweats. For some, these are transient nuisances. For others, they are debilitating: sleep-destroying, career-disrupting episodes that persist for years. Beyond the vasomotor symptoms, estrogen withdrawal accelerates age-related bone loss increasing fracture risk, alters blood lipid profiles, contributes to urogenital atrophy, and may affect cognition and mood. Although disease may not be the correct term for menopause, the phase exacts a physiological and psychological toll on many women. MHT emerged as a way to supplement estrogen with the goal of mitigating the debilitating vasomotor symptoms of menopause.
Replacing what is lost?
MHT is among the most effective therapies for symptoms emerging from menopause. The intervention has adopted several names: hormone replacement therapy (HRT), estrogen replacement therapy (ERT), or the currently preferred clinical term, menopausal hormone therapy (MHT). The alphabet soup reflects shifting attitudes more than shifting chemistry. While “HRT” and “ERT” imply restoring something lost, “MHT” may appear more neutral.
The pharmacology, however, is relatively straightforward. Estrogen is the primary active therapeutic agent. Many regimens incorporate a progesterone component to limit uterine endometrial hyperplasia.3 MHT is available in several forms: oral estradiol, transdermal patches and gels, vaginal rings and creams, and the older CEE (Premarin).4
The primary goal for MHT is the treatment of vasomotor symptoms. Estrogen is, by a wide margin, the most effective pharmacological intervention for hot flashes, reducing their frequency and severity by ≥75% in randomized trials. For the millions of women whose vasomotor symptoms impair their sleep, work, and quality of life, estrogen transforms their quality of life.
Beyond symptom relief, MHT/ERT has the well-documented ability to slow bone loss through osteoclast-mediated bone resorption. By preserving bone mineral density, estrogen MHT can reduce hip fracture risk by 1/3rd.
However, for decades, clinicians and researchers debated additional benefits: cardiovascular protection, reduction in colorectal cancer, improvements in mood and cognition. Some of these claims have been supported by evidence; others have not. The contested benefits, particularly cardiovascular protection, propelled MHT into one of the most consequential clinical controversies of the past half-century. In an attempt to settle the debate, the NIH’s Women’s Health Initiative (WHI) commissioned large, randomized controlled trials to evaluate the risks and benefits of MHT in post-menopausal women.
In the early 1990s, the NIH launched the WHI to study the impacts of MHT/HRT in, largely, post-menopausal women. To date, this remains one of the largest public clinical RCT efforts. Investigators anticipated positive results supporting widespread utilization of MHT. In 2002, the WHI’s trial of CEE+MPA in post-menopausal women was halted early. Contrary to expectations, MHT appeared to increase risks across the board.
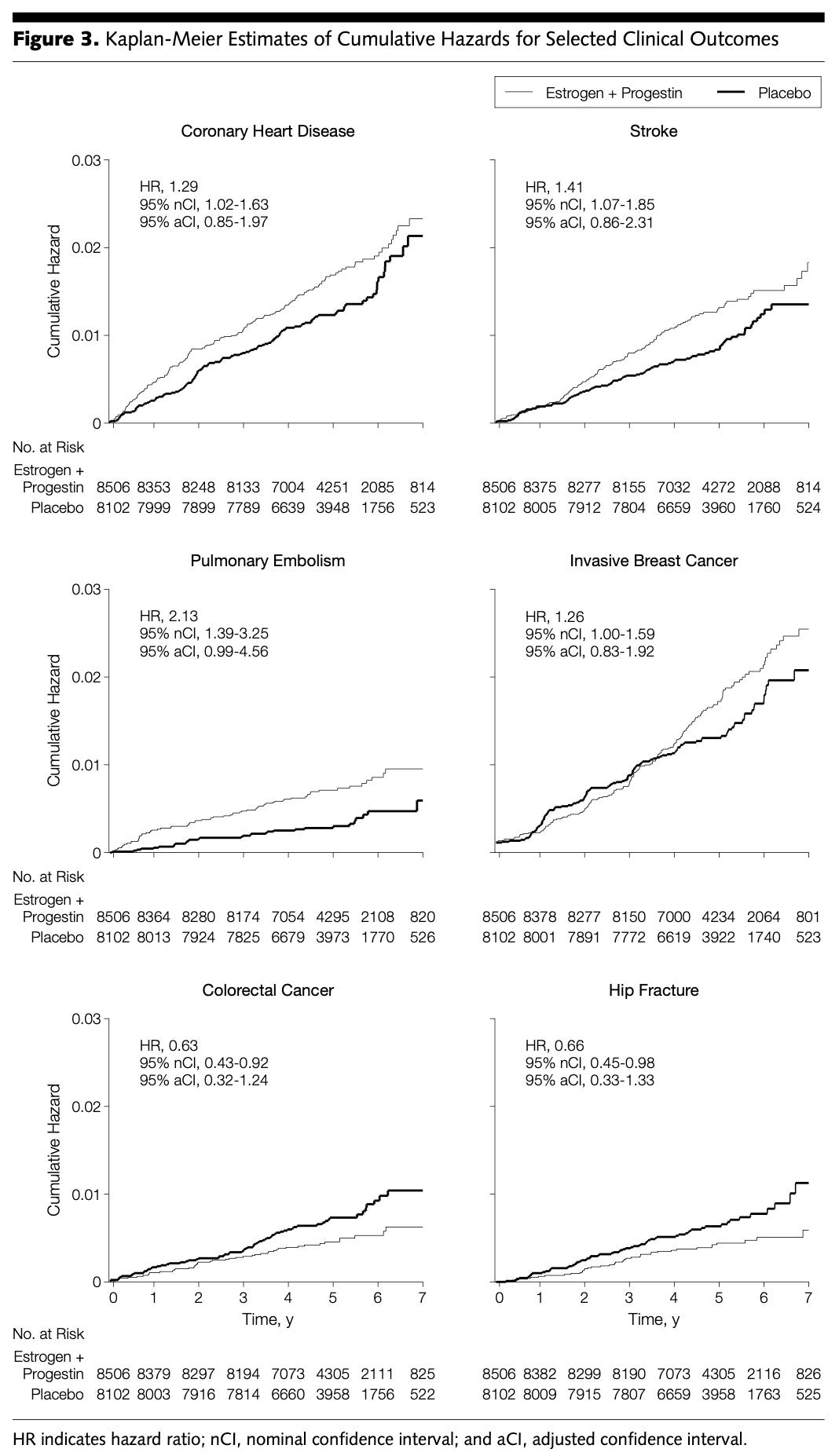
Summarized above in Figure 3 from the JAMA publication, MHT appeared to increase the risk of breast cancer, dementia, and cardiovascular disease. However, the trial did confirm the benefit of MHT in reducing fracture risk. The balance of risk led the FDA to apply a blanket black-box label to all estrogen containing MHT products.
Ripping the bandage
However, in February 2026, the FDA approved labeling changes to the first batch of menopausal hormone therapy products, removing risk statements about cardiovascular disease, breast cancer, and probable dementia from the boxed warning. This issue has been a pet-project of FDA Commissioner Marty Makary.5 A surgeon and public health researcher, Makary has gone on record calling the decades-long stigmatization of HRT “one of the greatest mistakes in modern medicine.”
A 2013 post-hoc reanalysis of the WHI (2002) results suggested that risks of MHT clustered largely in women >10 years post-menopause. Women 50-59 on MHT appeared to have reduced all cause mortality, but still observed increased cardiovascular and cancer risks. Across all groups, MHT appeared to reduce colorectal cancer and hip fracture risk. Therefore, MHT may provide symptomatic benefit during menopause and may be effective primary-prevention
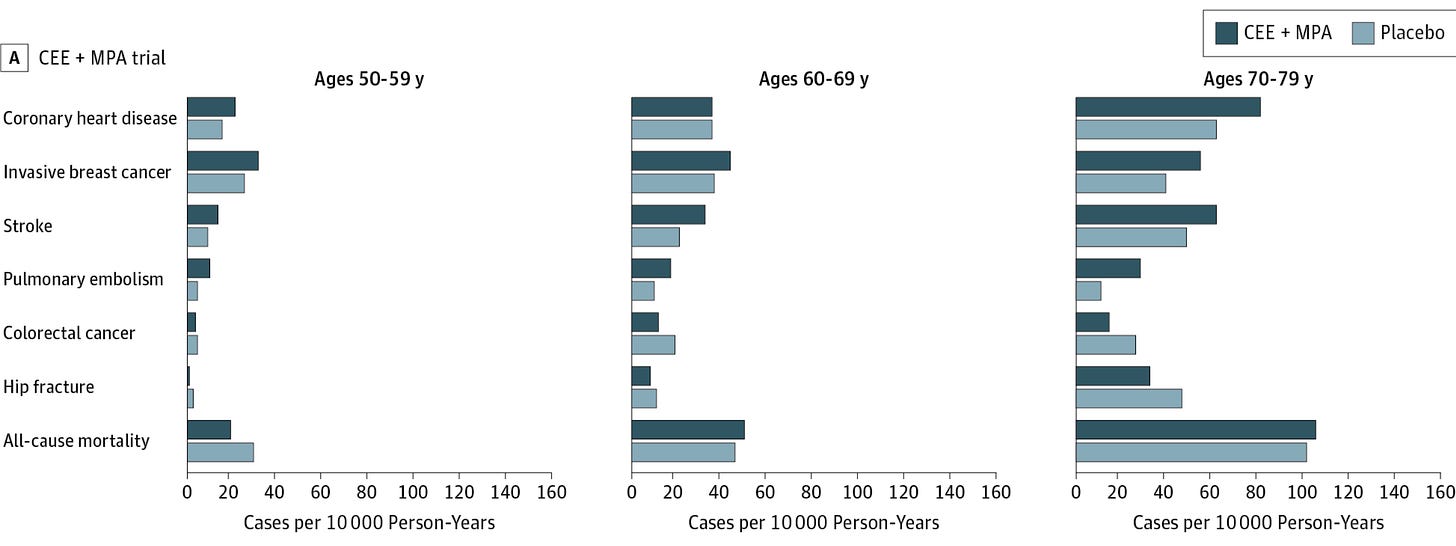
The reasoning, laid out in a JAMA Viewpoint authored by Makary, rested on four pillars.
Misapplication of the 2002 WHI results. The trial tested a specific oral regimen, CEE plus MPA, in a population averaging 63 years old implying many women were a decade post-menopausal. However, the warnings were applied to all formulations, all ages, all MHT indications. Makary argues this application was overly broad.
Age-stratified benefit. The age-stratified re-analyses reversed the risk-benefit calculus for younger women. For women initiating therapy within 10 years of menopause, the randomized data show reductions in all-cause mortality and fractures, with no apparent increase in cardiovascular events.
Newer, safer formulations. The formulations prescribed today are not the formulations the WHI tested. Transdermal estradiol patches and gels bypass the liver’s first-pass metabolism that is thought to drive the clotting risk of oral estrogens, as observed in some oral contraceptive formulations. Oral micronized progesterone has replaced the synthetic MPA that the WHI implicated in the breast cancer and cardiovascular risk increases. The WHI investigators themselves acknowledge they never tested these newer formulations.
Potential for broad impact. The black box created a chilling effect that harmed women. In 2020, there were 40 million American women aged 45-64, but only about 2 million received an MHT prescription. A generation of physicians trained after 2002 received little instruction in prescribing menopausal hormone therapy. In his MHT review, Lobo (2016) called them “the lost generation of trainees.” The result: an entire cohort of clinicians uncomfortable prescribing a menopause therapy that may be broadly beneficial.
The specific changes: cardiovascular, breast cancer, and dementia risk statements were removed from the Black Box Warning. The updated label will recommend initiation before age 60 or within 10 years of menopause onset. The endometrial cancer warning for estrogen-alone products, which the re-analyses did not overturn, remains. Risk information about cardiovascular disease and breast cancer stays in the label, but in the “Warnings and Precautions” section rather than as a Black Box Warning. Major MHT drug companies submitted proposed labeling changes at the FDA’s request.
Will a new label broaden access?
The relabeling matters because the Black Box was not merely informational—it was behavioral. A boxed warning is the most prominent warning signal the FDA can attach to a drug. It shapes formulary decisions, insurance coverage, malpractice exposure, and patient willingness to fill a prescription. For two decades, the Black Box on HRT products told physicians and patients, in the starkest possible terms, that this class of medication could cause heart attacks, breast cancer, and dementia. The nuance, that these risks applied primarily to older women starting a specific oral regimen years after menopause, was invisible on the label.
For the millions of women in their 40s and 50s experiencing debilitating vasomotor symptoms, the practical effects of relabeling could be transformative. The gap between the 40 million women who might benefit and the 2 million who currently receive therapy is not explained by clinical contraindications alone. Importantly, this gap - two decades of women who went through menopause with MHT support - won’t benefit from this relabeling change. These women, now in their 50s, 60s, and 70s have already preserved through menopause and experienced irreversible changes to their physical health, including the silent bone loss that will likely go undetected until fracture. Much of this impact appears explained by fear rooted in a warning that, for the intended population of younger menopausal women, that may be overstated given the magnitude of risk observed in the WHI.
In some ways, the MHT story has come full-circle. In the 1960s, drug companies sold estrogen as a cure-all for menopausal women. Evidence? Not much. Yet, estrogens were widely adopted among peri- and post-menopausal women. That, in turn, inspired the WHI which read out in 2002. As, Makary et al. argued, the FDA overgeneralized from WHI inaugurating a quarter-century of therapeutic nihilism. Now, with relabeling, the FDA attempts to restore a nuanced, population-specific understanding of HRT’s risks and benefits.
Perhaps this change represents a genuine correction. Or, perhaps, it is another pendulum swing back toward enthusiasm. Will patients benefit? It’s not totally clear. Ultimately, only rigorous evidence can inform the decision to initiate and continue MHT. Yet, given the diversity among MHT formulations and regimens, we still have many degrees of freedom to flex before we get a clear answer to the risk-benefit of MHT across all peri- and post-menopausal women.
How did we get here?
But, how did we get here? Why did the NIH sponsor the multi-decade Women’s Health Initiative? How did MHT become so popular such that over 1-in-4 postmenopausal women were taking at the turn of the century? And, how was it discovered in the first place?
The history of menopausal hormone replacement therapy is a long and fascinating road. I became interested in the origins of HRT after learning about how estrogen supplementation was developed as a front-line preventative for osteoporosis. The relabeling is the most recent chapter in a story that starts in the late 1700s, runs through the barnyard, catalyzed the emergence of a new discipline of medicine, and is still being written. As I see it, the story has five parts:
Part 1 - Reviving Estrogen Replacement Therapy
Part 2 - Organ Replacement
Part 3 - Hunt for the Hormone
Part 4 - Estrogen Rush
Part 5 - Weighing the Scales of MHT
After tracing the history of MHT, it’s not clear to me that we’ll ever have concise answers to the plethora of questions around MHT. The argument over whether medicine should provide MHT is possibly older than MHT itself. In many ways, MHT is a vestige of a different time in medicine where discoveries arrived and scaled rapidly on what we would now consider weak a evidentiary basis.
In some ways, the story of MHT reflects the difficulty of generating high quality evidence to inform patients and providers. The NIH paid for a large, decade-long randomized controlled trial of MHT with the Women’s Health Initiative. Yet, the answers remain unclear! Further, I believe that the story of MHT highlights the tension between speed and rigor. The pace of medical progress can accelerate, but the certainty we can have about the benefits and risks of a therapy necessarily diminish. Enthusiasm about the potential benefits bolstered by testimonials from patients and providers, amplified by the drugmakers themselves, brought MHT into the mainstream. Few interventions have been adopted as broadly as MHT with so little high-quality evidence. Yet, some people clearly benefit from MHT. And, the FDA has MHT slated for a grand comeback.
So, how did Estrogen Replacement Therapy develop in the first place?
The average age of natural menopause in the United States is 51 years. Premature menopause (before 40) and early menopause (40–45) affect approximately 1% and 5% of women, respectively.
This is contested. Some work suggests oopause is common across mammals. However, these data rely on observations from captive animals where reproduction behaviors can diverge from natural habitats.
For women with an intact uterus, a progestogen must be co-administered to prevent estrogen-driven endometrial hyperplasia, which can progress to endometrial cancer. The progestogen can be synthetic (medroxyprogesterone acetate, or MPA) or natural (oral micronized progesterone). Women without a uterus—roughly one-third of American women over 60, owing to the historically high rate of hysterectomy—can take estrogen alone.
Conjugated equine estrogens (CEE), marketed as Premarin, were first introduced by Ayerst in 1942. CEE is a complex mixture of at least ten estrogens, including estrone sulfate (~50%), equilin, and 17α-dihydroequilin, several of which are not found in humans.