Part 3: Isolating Estrogen
A putrid purification
By the end of the second decade of the twentieth century, the study of the ovary had arrived at a peculiar impasse: a great deal was known about the ovary, yet almost nothing could be done with the knowledge.
The popularity of Battey’s operation, the bilateral oophorectomy, left a generation of women in surgical menopause. However, this operation established the causal relationship between ovarian failure and menopause first postulated by Percival Pott in the late 18th century and later described explicitly by Adolf Hegar in Die Castration der Frauen. The ovary had been established as a valuable gland for reproduction and health.
But, medicine couldn’t do much for women with ovarian failure. The pioneering surgeon Robert Tuttle Morris attempted ovarian transplants. Adventurous physicians prescribed organ extracts for menopausal symptoms. It’s unclear the degree to which these benefited patients. Scientists needed to identify, purify, and produce the ovarian hormone before physicians could treat patients at scale.
The Dawn of Endocrinology
Continued interest in the functions of the ovary intersected with the dawn of a discipline focused on the study of internal secretions: Endocrinology. In what became the “Heroic age of Endocrinology,” pioneering physicians and scientists refined and purified the organ extracts of crude “Organotherapy” into their biochemical essences.
The quest to refine the secretions of organ glands started in the late 1800s with the discovery of adrenaline. In 1895, George Oliver and Edward Albert Schäfer isolated adrenaline from adrenal glands.1 Unrealized in the moment, their adrenal extract was the first hormone to be characterized, later purified by the Japanese chemist Jokichi Takamine in 1901, and synthesized by Friedrich Stolz and Henry Drysdale Dakin, independently, in 1904.
In 1905 the physiologist Ernest Starling refined the concept of “internal secretion,” as first exemplified by Berthold’s experiments transplanting rooster testes, endogenous action substance, or “hormone,” from the Greek verb ‘hormao’ meaning to excite or set in motion. With William Bayliss, Starling had recently discovered “secretin” in 1902, a substance that the intestine released into the blood to activate the pancreas. This discovery overturned the long-held belief that the nervous system activated the pancreas and digestive system in response to food intake.
The discovery of most endocrine hormones followed a fairly similar framework.
Identification of the Source - What organ produces the functional secretion? Adrenaline, for example, is produced in the medulla (inner layer) of the adrenal gland
Detection Method (Bioassay) - How do you reliably detect the activity of the substance? For Oliver and Schäfer, this was heart rate and blood pressure. Administration of adrenal extract raised the heart rate and blood pressure of humans and dogs
Extract Preparation - How do you extract the active substance from the gland? Typically, this involved mechanical disruption followed by dissolution of the gland in water or various organic solvents (oil, ether, petroleum fractions, etc.)
Purification and Characterization - Can you isolate a single molecule responsible for the activity? Generally, to prove identity, extracts are refined through fractionation, precipitation, and recrystallization until the substance is homogenous and potent in turn isolating the active component. Then, elemental analysis could be used to determine a molecular formula. Finally, for small, organic molecules, identity can be confirmed by chemical synthesis of the molecule from a simpler components. If the synthesized molecule is identical to the extract in elemental composition, activity, and potency, then the identity and structure of the hormone has been fully elucidated2
For scientists focused on reproductive biology, like Edgar Allen, the hormone concept catalyzed a search for the essential hormones of the reproductive glands. Decades of clinical observation and experimental work — the transplantation experiments of Knauer and Halban, the clinical reports of oral ovary organotherapy, and Tuttle’s ovarian transplants — had suggested that the ovary must produce something essential for reproduction and female health. Now there was a scientific framework in which to ground the search. The belief emerged that ovary must secrete a hormone into the blood that controls reproduction; the disappearance of that hormone explains the loss of menses and the onset of menopausal symptoms; and the replacement of that hormone should restore what the ovary lost in menopause. This logic, speculative yet elegant, turned out substantially correct.
Beneath this seemingly straightforward picture lay complications that would take decades to untangle. The earliest researchers made a fatally simplifying assumption: that they were hunting a single “ovarian hormone,” a unified substance responsible for all ovarian actions. In reality, the ovary produces multiple hormones with distinct roles and timing. Fraenkel’s 1901 demonstration that the corpus luteum was essential for pregnancy had already hinted at this complexity, suggesting that two parts of the ovary - the follicle and the corpus luteum - might secrete different substances.3 But the distinction between estrogens and progesterone would perplex investigators for years. The assumption of unity — one gland, one hormone — was a useful simplification that would eventually have to be abandoned.
The hunt for the ovarian hormone
By the early 1920s, the endocrine function of the ovary had been well established. However, the ovarian hormone remained mysterious. The ovary, in particular, is a complex endocrine organ. In the ovary, follicles grow and mature the reproductive eggs inside the follicular capsule. After ovulation, release of the egg, the follicle retreats into a mass of scar-like tissue known as the corpus luteum before regression. In what sub-structure of the ovary is ovarian hormone produced? Is the hormone in the follicles? The corpus luteum? The interstitial tissue? This was the question that Edward Doisy and Edgar Allen sought to answer.
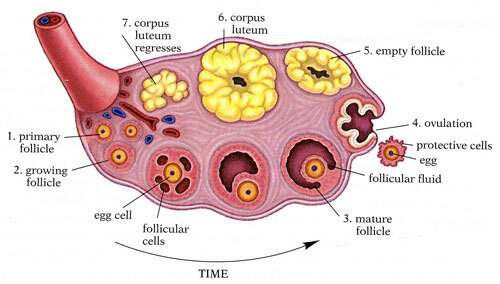
In 1923, Allen and Doisy identified the follicular fluid, in the mature follicles, as the source of the primary ovarian hormone.4 Three key developments enabled this work.
Source: Hog Follicles - the ovaries of hogs had larger ovarian follicles than common laboratory species (rats, mice) and were readily available as byproducts of the meatpacking industry. Doisy painstakingly aspirated the follicular fluid (liquor folliculi) from the ovaries that Allen would use in his animal assays
Assay: Juvenile Mice - Edgar Allen developed an assay to assess ovarian activity in an immature mouse. A zoologist by training, Allen systematically classified the stages of the estrous cycle by the cell types present in vaginal smears: nucleated epithelial cells in proestrus, cornified (keratinized, non-nucleated) epithelial cells at estrus, and leukocytes predominating in diestrus. His key observation was that vaginal cornification, the appearance of flat, keratinized cells shed from the vaginal lining, reliably indicated the estrus phase and, by extension, peak estrogenic stimulation.5 Administration of the follicular extract to the juvenile mouse accelerated the sexual maturation, as indicated by vaginal cornification, of the mice
Assay: Ovariectomized Juvenile Mice - in his doctoral work, Allen developed the ovariectomized mouse as a model for studying reproduction. Removing the ovaries from immature mice would arrest sexual development and ensure that endogenous ovarian hormones couldn’t contaminate the result. Working with Doisy, Allen inject supplemental follicular fluid into the immature, ovariectomized mice to restore sexual maturation and cornification of the lining of the vaginal walls.
Together, these developments unlocked the ability of Allen and Doisy to assay the sub-compartments of the ovary to determine what portion held the ovarian hormone. A competing theory was that the corpus luteum, the remnant of the post-ovulatory follicle, contained the ovarian hormone. Allen and Doisy’s mouse assays effectively ruled out the corpus luteum as the source.6 Only the fluid of the follicle rescued sexual development on ovariectomized juvenile mice. This assay became known as the Allen-Doisy test. Allen and Doisy’s work pinpointed the location of the ovarian hormone, but their characterization failed to produce the purified substance.
This inability to isolate the pure ovarian hormone was not unexpected. Incredibly potent, endogenous hormones exist in their glands at very low concentrations. For example, during the characterization of thyroxine, the thyroid hormone, 3 tons of pig thyroids produced only 33 grams of pure thyroxine.7 To identify the ovarian hormone, Allen and Doisy needed a new source: abundant, accessible, and ideally concentrated.
Along comes… a reliable pregnancy test?
In the mid-1920s, two German gynecologists, Selmar Aschheim and Bernhard Zondek, were on a quest to solve a problem that now seems mundane: reliably determine pregnancy. Prior to Aschheim and Zondek’s work, physicians primarily diagnosed pregnancy with physical palpations and only well into the first trimester. Moreover, distinguishing an early pregnancy from a tumor or large fibroid sometimes proved challenging. To illustrate the point, Aschhem and Zondek reported that physicians would occasionally diagnose a uterine tumor and begin operation only to find a pregnant uterus.8
The core assumption of Aschheim and Zondek’s work was that the prior observation that many hormonal substances appeared massively enriched in the blood pregnant women. If these hormones were present, Aschheim and Zondek assumed the hormones must be excreted in some fashion.9 And, what do pregnant women excrete? Breast milk and urine. So, Aschheim and Zondek assayed both breast milk and urine for their ability to observe ovarian enlargement, appearance of a corpus luteum, uterine enlargement, and follicle maturation in juvenile mice. To their delight, only the urine of pregnant women drove the signs of sexual and follicular maturation. In 1927, after testing 2000 samples of urine, Aschheim and Zondek reported a 98.9% success rate in positively determining pregnancy!
This method quickly became known as the Aschheim-Zondek Test (A-Z test) for pregnancy. It would later be adapted to rabbits (Freidman test) and then to ovulation in African clawed frogs (Hogben test) and eventually sperm production in male toads (Galli Mainini). The amphibian-based tests remained popular for decades until the development of an immunoassay as both tests could be completed quickly and did not require termination of the animal, in contrast to the A-Z and Freidman tests. What incredibly well conserved hormone drives these transformations? The chorionic gonadotrophin (hCG) from the placenta — not an ovarian hormone.
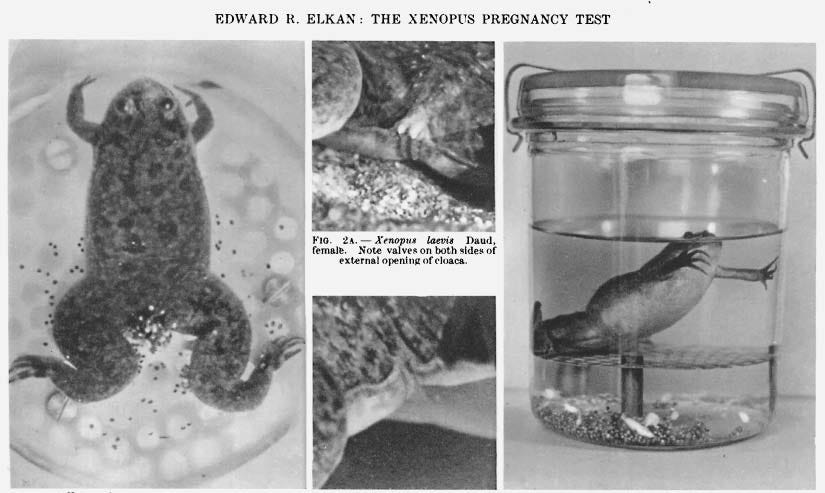
So what does a pregnancy test have to do with isolating the elusive ovarian hormone? Well, in their seminal work, Aschheim and Zondek reported that the urine of pregnant women had concentrated activity that mirrored Allen and Doisy’s ovarian hormone.10 That observation transformed Allen and Doisy’s hormone hunt.
Isolating estrone
Previously limited to painstaking follicular fluid extraction from porcine ovaries, in Aschheim and Zondek’s report, Allen and Doisy found a cheap, abundant feedstock: urine, from pregnant women. Unbeknownst to the investigators, the placenta, not the ovary, produced the ovarian hormone. But, the urine was active. So, Allen and Doisy did what the early hormone heroes did, collect and process an unfathomable amount of hormone enriched waste.
Doisy set about collecting urine from pregnant women the obstetrics clinic at Washington University St. Louis. At every step, the Allen-Doisy assay guided the purification: extract, fractionate, test each fraction in the mouse, pursue the active fraction, repeat. The work was as laborious as it was unglamorous.
In an autobiographical feature, Doisy recalls that at one point in the project a policeman searching for bootlegged alcohol pulled over his Model T. To the policeman’s disappointment, Doisy’s collection of gallon bottles contained naught but the urine of women, pregnant.
From hundreds of gallons of urine, Doisy extracted the estrogen producing pure white crystals of estrone, the first estrogen. Doisy named the compound "theelin," from the Greek thely, meaning female. Simultaneously, German chemist Adolf Butenandt isolated estrone following a similar process, allegedly after hearing about a presentation of Doisy’s at the 1929 International Physiologic Congress in Boston, although the order of events is disputed.11 Butenandt, too, had used pregnant women's urine as his starting material, guided by the same bioassay framework. He called his compound "Follikelhormon," follicular hormone. The simultaneous discovery of estrone, the first estrogen, created a chaotic nomenclature that required an international conference to resolve.
Yet estrone, it turned out, was only the first of three natural estrogens to be identified, and it was neither the most potent nor the physiologically dominant one. The order in which the three classical estrogens were isolated is counterintuitive, a paradox that reveals much about the practical constraints of early hormone chemistry.
Hormonal Babel
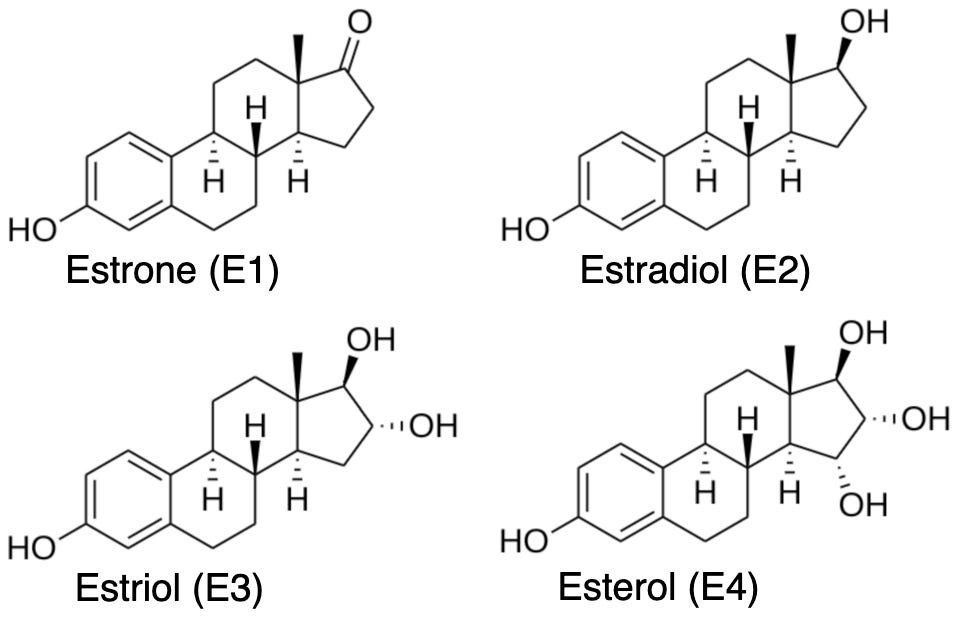
Guy Frederic Marrian, working at University College London, isolated a second estrogen from human pregnancy urine around 1930. This compound, which Marrian called “trihydroxyoestrin,” had three hydroxyl groups, or estriol. It was the weakest of the three natural estrogens, roughly one-tenth the potency estradiol. It is, however, the dominant estrogen of pregnancy, produced in vast quantities by the fetoplacental unit. Doisy and Butenandt isolated estrone, the oxidized product, as a consequence of their purification process.
The most potent natural estrogen, the one whose decline defines menopause. was the last to be isolated. In 1935, Doisy’s group extracted estradiol from sow ovaries. The yield was breathtakingly meager: twelve milligrams of crystalline estradiol from four tons of porcine ovaries.12 The most potent of the three natural estrogens, estradiol is the principal estrogen secreted by the ovary in premenopausal women and the reference standard against which all other estrogens are measured.
The rapid, competitive, multi-laboratory nature of the estrogen isolation race produced a predictable consequence: nomenclatural chaos. Different laboratories assigned different names to what were often the same molecule, creating a literature so confused that even specialists struggled to compare results across publications.
Doisy in St. Louis called his compounds theelin (estrone), theelol (estriol), and dihydrotheelin (estradiol). Butenandt in Göttingen used Follikelhormon, marketed as Progynon. Ernst Laqueur’s group in Amsterdam, which isolated the same compound in 1930, called it menformon. In Geneva, Girard used the name folliculin. Marrian in London referred to estriol as trihydroxyoestrin.13
These were not different substances: they were the same molecules, independently discovered and independently named. The Babel hindered both scientific communication and clinical standardization.
The confusion was diminished by the International Conference on the Standardization of Sex Hormones in 1935. Here the pioneers of estrogen established the nomenclature that persists to this day: estrone for the hydroxyketone (E1), estradiol for the diol (E2), estriol for the triol (E3), systematic names derived from the functional groups on the steroid nucleus.14
Bringing estrogens to patients
Shortly after the reports of isolation, early pharmaceutical companies released a variety of preparations: Amniotin, Progynon, Theelin, Emmenin, Folliculin, and Oestroform. These preparations enriched estrone from the urine of pregnant women. These estrone preparations were naturally supply constrained. Where else could one find a potent and near limitless source of estrogen to treat menopause?
Chemical synthesis, perhaps? The structure of estrone had been elucidated by Budenandt in 1930. But, 1930s era chemical synthesis was not yet advanced enough to create large quantities of estrogens from the available precursors. In fact, it wasn’t until 1946 that Wiesbader and Filler used synthetic estradiol15 to treat menopausal symptoms, finally validating the identity of the key active component of ovarian extracts.
If not synthesis, what might unlock estrogen at the scale needed to treat the world’s women? Where might we find a truly limitless source of estrogen? Once again, the search takes us back to the barnyard.
A different adrenal extract preparation, epinephrine, was prepared by in 1897 by John Jacob Abel, one of the founders of modern pharmacology.
For peptide and protein hormones, this process can be considerably more complex
Medvei, 1993, pp. 202–203
The ASU Embryo Project has a helpful and more in-depth summary
Medvei, 1993, p. 219
The corpus luteum does produce an important hormone: progesterone, “the implantation hormone” (pro- gest-ation horm-one). Corner and Allen would isolate the other ovarian hormone, “the implantation” hormone progesterone, six years later
Aschheim and Zondek. “Die Schwangerschaftsdiagnose aus dem Harn Durch Nachweis des Hypophysenvorderlappenhormons.” Klin Wochenschr 7, 1404–1411 (1928). I machine translated this into English as my German isn’t great, so I’m moderately confident this example is correct.
This is true, but a fortuitous assumption as many hormones are modified, degraded, or inactivated before terminal excretion!
The Aschheim-Zondek test detects gonadotropin. Specifically, human chorionic gonadotropin, produced by the placenta during pregnancy. When hCG-rich urine is injected into an immature mouse, the hCG stimulates the mouse's own immature ovaries to produce estrogen endogenously. The mouse's own estrogen and the estrogen in the urine, both drive the vaginal cornification and uterine growth observed in the test. The A-Z test is only considered positive when ovaries are enlarged with corpus luteum present, hallmarks of ovulation, not the secondary sexual maturation observed from the ovulation induced follicle hormone production. The A-Z test is repeated if uterine growth and vaginal cornification are observed without markers of ovulation.
In 1930, Adolf Butenandt determined the molecular structure of estrone, in turn discovering the first steroid hormone. Isolation and characterization of steroid hormones proved to be a tremendously productive line of research. He continued his work with steroid hormones by isolating progesterone and testosterone soon after. In 1939, Butenandt won the Nobel Prize in Chemistry for his work isolating and characterizing steroid hormones including estrone. The Nazi government prohibited his acceptance, so he did not formally accept the award from 1949. Doisy would go on to win a Nobel Prize for the isolation and purification of vitamin K.
Medvei, 1993, p. 220
Partial synthesis of estrone from cholesterol was achieved in 1939. But, total synthesis of estrone wasn’t achieved until 1948 by Anner and Miescher.

